
Studies showed the vaccine to be about 75% effective in blocking the disease. LYMErix, manufactured by what was then SmithKline Beecham, induced patients to produce antibodies that a tick would absorb from its victim’s blood and that neutralized Borrelia burgdorferi. (Rapid viral mutation is a main reason that scientists have yet to develop a vaccine against HIV.) What’s more, the bacterium “has virtually no machinery to survive mutation,” Klempner says, so that researchers can identify and stick with antibodies that eradicate the bacterium rather than having to keep up with mutated versions. That lag, he notes, provides time for antibodies to get into the tick’s gut and prevent transmission of the bacterium before it sets off an infection in the human. 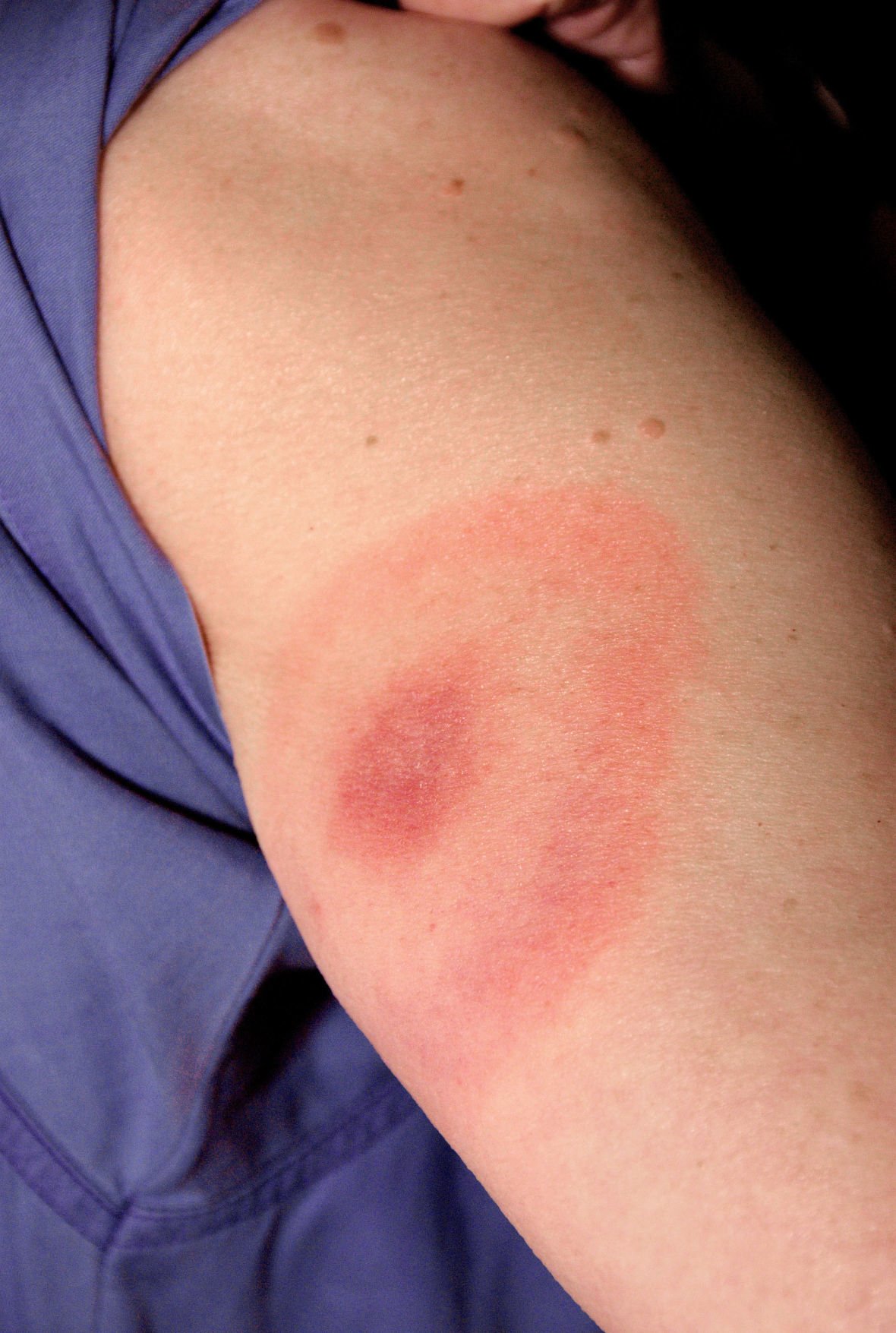
But when a black-legged tick latches onto someone, it takes at least 36 hours to pass along the Lyme-causing bacterium, Borrelia burgdorferi, says Gary Wormser, MD, chief of the Division of Infectious Diseases at New York Medical College. The science of stopping LymeĬreatures that spread diseases through bites - such as mosquitos that transmit malaria and raccoons that transmit rabies - usually deliver the virus, bacteria, or parasite instantly. The new efforts build on the basic strategy of those vaccines. “In terms of difficulty from a biological and medical standpoint, it was relatively straightforward.” “It was not very difficult to develop,” Klempner recalls of the vaccine, Imulym, made by Pasteur Merieux Connaught but never brought to market. Klempner knows that well, as he worked on one of two Lyme vaccines that showed strong clinical trial results in the 1990s. The lack of a human version has little to do with the science of making one. If any of these products are eventually approved by the Food and Drug Administration (FDA), it would put humans nearly on par with dogs they can get four Lyme vaccines made just for them. Researchers at the Yale School of Medicine report promising results from animal studies with a vaccine that teaches the body to recognize tick saliva and send an immune response to the site of the bite before the bacterium is transmitted.The medication developed by Klempner and colleagues at MassBiologics, which they call a vaccine alternative because it directly delivers a monoclonal antibody rather that spurring development of multiple antibodies in patients, is aiming for a phase 2-3 human trial next spring.A vaccine developed by Pfizer and Valneva, which induces the development of antibodies in people to stop the disease-causing bacterium within ticks, entered a phase 3 human trial last month.The current projects come two decades after the first human Lyme vaccine, LYMErix, was pulled from the market because of factors that researchers believe their new products will overcome. The disease sets off a series of escalating symptoms, starting with a red rash, fatigue, fever, headache, and a mild stiff neck, and sometimes escalating if left untreated to serious complications to joints (arthritis), the heart, and the nervous system. Targeting ticks to prevent Lyme rather than fighting off an infection after it begins is the fundamental strategy behind three projects that aim to create the only medication to prevent the disease in people. “The patient provides the vehicle to deliver the antibody to the infected tick.” Klempner, MD, a professor of medicine at UMass Chan. “We are trying to deliver a medicine to a tick so that it doesn’t pass on the infection to the patient,” says Mark S. They injected the antibody into humans so that people can transmit the antibody to ticks that bite them. That was the task taken on by a group of researchers at MassBiologics of UMass Chan Medical School (UMass Chan) in Massachusetts: They developed a way to get an antibody into ticks to neutralize the bacterium that causes Lyme disease, the most common vector-born disease in the United States.īut no, the researchers did not give shots to ticks.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
